Search
- Page Path
- HOME > Search
Original Article
- Clinical Study
- Development of a Non-Invasive Liver Fibrosis Score Based on Transient Elastography for Risk Stratification in Patients with Type 2 Diabetes
- Chi-Ho Lee, Wai-Kay Seto, Kelly Ieong, David T.W. Lui, Carol H.Y. Fong, Helen Y. Wan, Wing-Sun Chow, Yu-Cho Woo, Man-Fung Yuen, Karen S.L. Lam
- Endocrinol Metab. 2021;36(1):134-145. Published online February 24, 2021
- DOI: https://doi.org/10.3803/EnM.2020.887
- 4,503 View
- 132 Download
- 6 Web of Science
- 6 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
In non-alcoholic fatty liver disease (NAFLD), transient elastography (TE) is an accurate non-invasive method to identify patients at risk of advanced fibrosis (AF). We developed a diabetes-specific, non-invasive liver fibrosis score based on TE to facilitate AF risk stratification, especially for use in diabetes clinics where TE is not readily available.
Methods
Seven hundred sixty-six adults with type 2 diabetes and NAFLD were recruited and randomly divided into a training set (n=534) for the development of diabetes fibrosis score (DFS), and a testing set (n=232) for internal validation. DFS identified patients with AF on TE, defined as liver stiffness (LS) ≥9.6 kPa, based on a clinical model comprising significant determinants of LS with the lowest Akaike information criteria. The performance of DFS was compared with conventional liver fibrosis scores (NFS, FIB-4, and APRI), using area under the receiver operating characteristic curve (AUROC), sensitivity, specificity, positive and negative predictive values (NPV).
Results
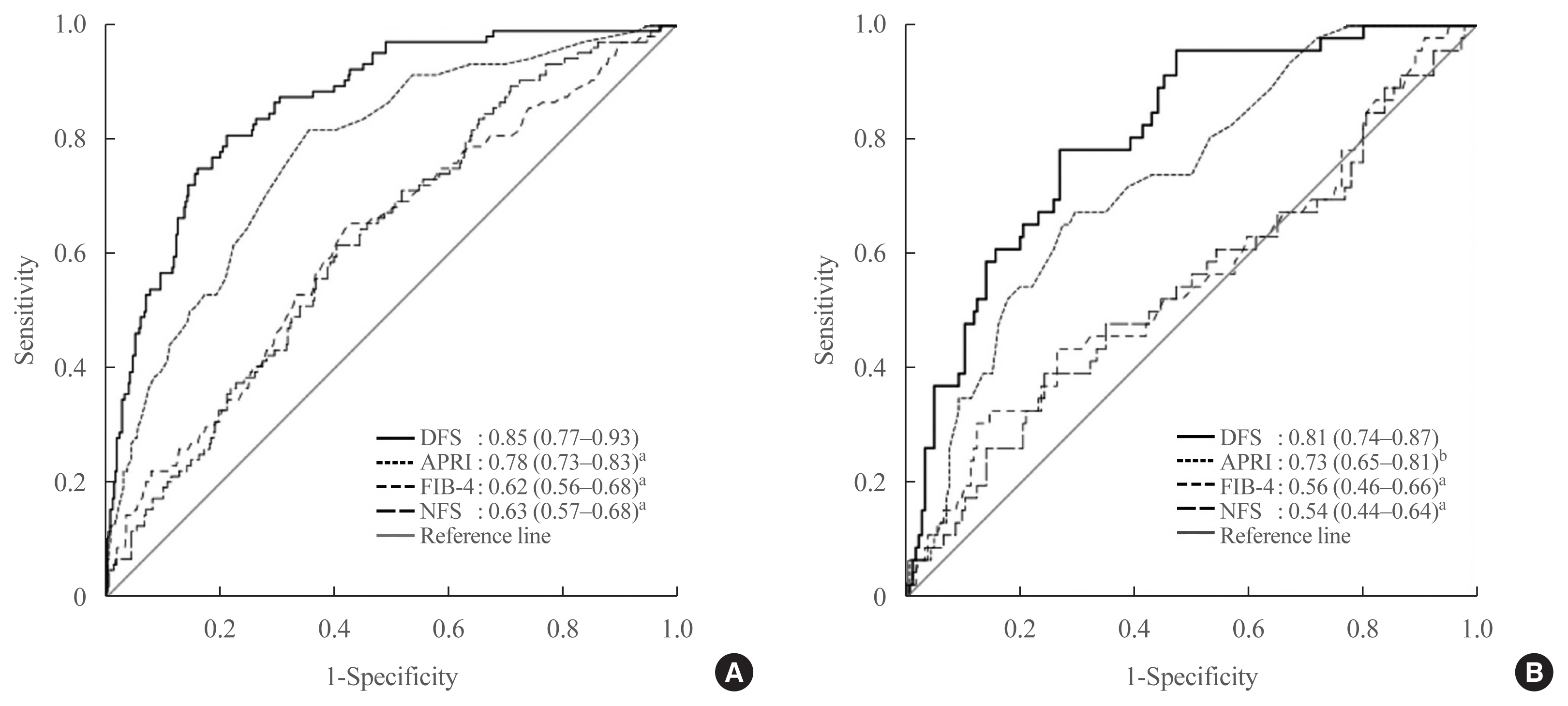
DFS comprised body mass index, platelet, aspartate aminotransferase, high-density lipoprotein cholesterol, and albuminuria, five routine measurements in standard diabetes care. Derived low and high DFS cut-offs were 0.1 and 0.3, with 90% sensitivity and 90% specificity, respectively. Both cut-offs provided better NPVs of >90% than conventional fibrosis scores. The AUROC of DFS for AF on TE was also higher (P<0.01) than the conventional fibrosis scores, being 0.85 and 0.81 in the training and testing sets, respectively.
Conclusion
Compared to conventional fibrosis scores, DFS, with a high NPV, more accurately identified diabetes patients at-risk of AF, who need further evaluation by hepatologists. -
Citations
Citations to this article as recorded by- Implementation of a liver health check in people with type 2 diabetes
Kushala W M Abeysekera, Luca Valenti, Zobair Younossi, John F Dillon, Alina M Allen, Mazen Noureddin, Mary E Rinella, Frank Tacke, Sven Francque, Pere Ginès, Maja Thiele, Philip N Newsome, Indra Neil Guha, Mohammed Eslam, Jörn M Schattenberg, Saleh A Alqa
The Lancet Gastroenterology & Hepatology.2024; 9(1): 83. CrossRef - Sequential algorithm to stratify liver fibrosis risk in overweight/obese metabolic dysfunction-associated fatty liver disease
Chi-Ho Lee, David Tak-Wai Lui, Raymond Hang-Wun Li, Michele Mae-Ann Yuen, Carol Ho-Yi Fong, Ambrose Pak-Wah Leung, Justin Chiu-Man Chu, Loey Lung-Yi Mak, Tai-Hing Lam, Jean Woo, Yu-Cho Woo, Aimin Xu, Hung-Fat Tse, Kathryn Choon-Beng Tan, Bernard Man-Yung
Frontiers in Endocrinology.2023;[Epub] CrossRef - Non-Invasive Measurement of Hepatic Fibrosis by Transient Elastography: A Narrative Review
Luca Rinaldi, Chiara Giorgione, Andrea Mormone, Francesca Esposito, Michele Rinaldi, Massimiliano Berretta, Raffaele Marfella, Ciro Romano
Viruses.2023; 15(8): 1730. CrossRef - Metabolic dysfunction-associated fatty liver disease — How relevant is this to primary care physicians and diabetologists?
Chi-Ho Lee
Primary Care Diabetes.2022; 16(2): 245. CrossRef - Non‐alcoholic fatty liver disease and type 2 diabetes: An update
Chi‐H Lee, David TW Lui, Karen SL Lam
Journal of Diabetes Investigation.2022; 13(6): 930. CrossRef - Ultrasound-Based Hepatic Elastography in Non-Alcoholic Fatty Liver Disease: Focus on Patients with Type 2 Diabetes
Georgiana-Diana Cazac, Cristina-Mihaela Lăcătușu, Cătălina Mihai, Elena-Daniela Grigorescu, Alina Onofriescu, Bogdan-Mircea Mihai
Biomedicines.2022; 10(10): 2375. CrossRef
- Implementation of a liver health check in people with type 2 diabetes


 KES
KES

 First
First Prev
Prev



